Spanish researchers pave way for fair play in global Covid testing and research
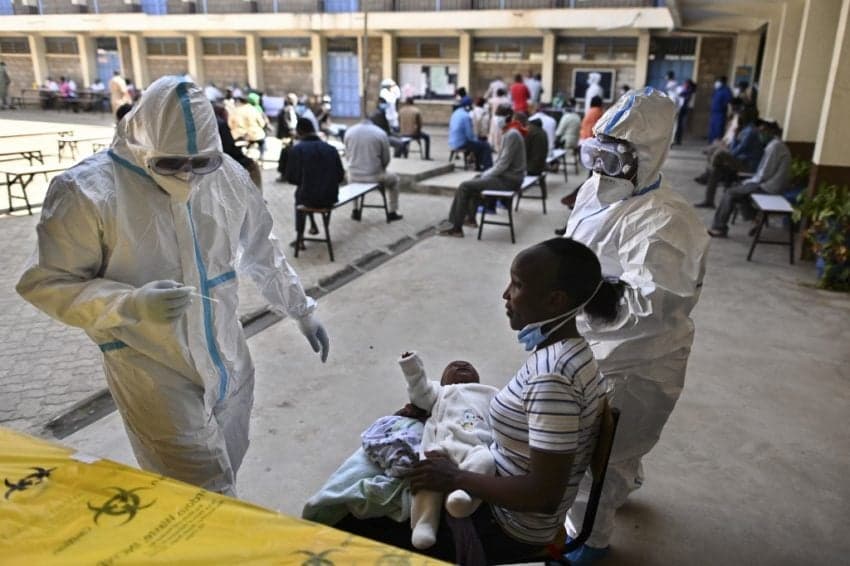
Spain’s top research institution - the CSIC - reached a licencing deal Tuesday that paves the way for its Covid-19 antibody test to be produced more cheaply in developing countries.
The World Health Organisation described the accord as the first transparent, global, non-exclusive licence for a Covid-19 health tool, that should help towards correcting the "devastating global inequity" in access.
The deal brings the Spanish National Research Council CSIC together with the global Medicines Patent Pool (MPP) and the WHO's Covid-19 Technology Access Pool (C-TAP) knowledge-sharing platform.
"The aim of the licence is to facilitate the rapid manufacture and commercialisation of CSIC's Covid-19 serological test worldwide," the WHO said.
The test effectively detects anti-SARS-CoV-2 antibodies developed in response to either a Covid-19 infection or a vaccine.
CSIC, one of Europe's main public research institutions, will provide the MPP or prospective licencees with know-how and training.
WHO chief Tedros Adhanom Ghebreyesus described the licence, which will be royalty-free for low and middle-income countries, as "the kind of open and transparent licence we need to move the needle on access during and after the pandemic".
He added: "I urge developers of Covid-19 vaccines, treatments and diagnostics to follow this example and turn the tide... on the devastating
global inequity this pandemic has spotlighted."
C-TAP was founded in May 2020 as a platform for developers of Covid-19 tools to share knowledge and intellectual property.
Set up during the scramble for Covid vaccines and treatments, the health technology repository was first suggested by Costa Rican President Carlos Alvarado.
The information pool was intended as a voluntary global bank for IP and open-sourced data as part of a common front against the new coronavirus.
However, as it turned out, rival pharmaceutical companies have largely kept their findings to themselves rather than sharing them as global public goods.
Tuesday's deal "shows that solidarity and equitable access can be achieved", said Alvarado.
CSIC president Rosa Menéndez said she hoped the move would serve as an example for other research organisations.
'Preposterous' tests hoarding
The medical charity Doctors Without Borders (MSF) said the test could quantify three different types of antibodies -- and crucially, differentiate vaccinated people from those with natural Covid infection.
"This feature will become very important for measuring the number of Covid-19 cases in countries and the impact of control measures," it said.
In welcoming CSIC's move, MSF diagnostics adviser Stijn Deborggraeve said it was "preposterous" in a global pandemic that tests were being monopolised by "a handful of privileged people and countries".
The Geneva-based MPP is a UN-backed international organisation that works to facilitate the development of medicines for low- and middle-income nations.
The antibody test licensing accord is the third Covid-related deal that the global pool has struck in a month.
Last week, the MPP reached an agreement with US pharmaceutical giant Pfizer to make its prospective antiviral Covid-19 pill available more cheaply in 95 low- and middle-income countries via sub-licensing to generic drug manufacturers.
The MPP signed a similar deal last month with Pfizer's US rival Merck for its prospective oral antiviral medicine molnupiravir.
Comments
See Also
The World Health Organisation described the accord as the first transparent, global, non-exclusive licence for a Covid-19 health tool, that should help towards correcting the "devastating global inequity" in access.
The deal brings the Spanish National Research Council CSIC together with the global Medicines Patent Pool (MPP) and the WHO's Covid-19 Technology Access Pool (C-TAP) knowledge-sharing platform.
"The aim of the licence is to facilitate the rapid manufacture and commercialisation of CSIC's Covid-19 serological test worldwide," the WHO said.
The test effectively detects anti-SARS-CoV-2 antibodies developed in response to either a Covid-19 infection or a vaccine.
CSIC, one of Europe's main public research institutions, will provide the MPP or prospective licencees with know-how and training.
WHO chief Tedros Adhanom Ghebreyesus described the licence, which will be royalty-free for low and middle-income countries, as "the kind of open and transparent licence we need to move the needle on access during and after the pandemic".
He added: "I urge developers of Covid-19 vaccines, treatments and diagnostics to follow this example and turn the tide... on the devastating
global inequity this pandemic has spotlighted."
C-TAP was founded in May 2020 as a platform for developers of Covid-19 tools to share knowledge and intellectual property.
Set up during the scramble for Covid vaccines and treatments, the health technology repository was first suggested by Costa Rican President Carlos Alvarado.
The information pool was intended as a voluntary global bank for IP and open-sourced data as part of a common front against the new coronavirus.
However, as it turned out, rival pharmaceutical companies have largely kept their findings to themselves rather than sharing them as global public goods.
Tuesday's deal "shows that solidarity and equitable access can be achieved", said Alvarado.
CSIC president Rosa Menéndez said she hoped the move would serve as an example for other research organisations.
'Preposterous' tests hoarding
The medical charity Doctors Without Borders (MSF) said the test could quantify three different types of antibodies -- and crucially, differentiate vaccinated people from those with natural Covid infection.
"This feature will become very important for measuring the number of Covid-19 cases in countries and the impact of control measures," it said.
In welcoming CSIC's move, MSF diagnostics adviser Stijn Deborggraeve said it was "preposterous" in a global pandemic that tests were being monopolised by "a handful of privileged people and countries".
The Geneva-based MPP is a UN-backed international organisation that works to facilitate the development of medicines for low- and middle-income nations.
The antibody test licensing accord is the third Covid-related deal that the global pool has struck in a month.
Last week, the MPP reached an agreement with US pharmaceutical giant Pfizer to make its prospective antiviral Covid-19 pill available more cheaply in 95 low- and middle-income countries via sub-licensing to generic drug manufacturers.
The MPP signed a similar deal last month with Pfizer's US rival Merck for its prospective oral antiviral medicine molnupiravir.
Join the conversation in our comments section below. Share your own views and experience and if you have a question or suggestion for our journalists then email us at [email protected].
Please keep comments civil, constructive and on topic – and make sure to read our terms of use before getting involved.
Please log in here to leave a comment.