Spain probes death of Marbella teacher after AstraZeneca jab
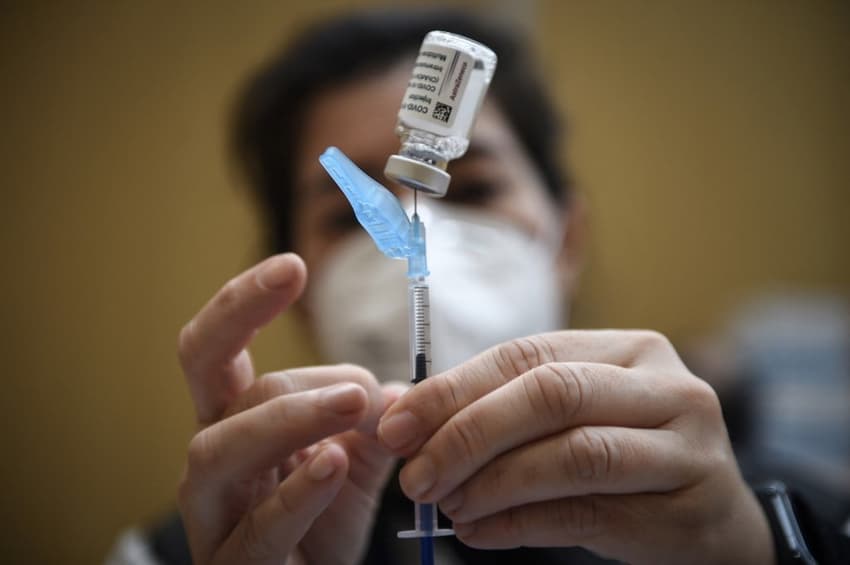
Spanish health officials said Wednesday they were investigating three cases of people who suffered from thrombosis after receiving the AstraZeneca vaccine, one of whom died, AFP has reported.
The announcement by Spain's AEMPS medicines agency came two days after the government suspended use of the vaccine for at least a fortnight as a precautionary measure.
The three cases occurred between late Monday and early Tuesday.
Local press reports said the person who died was a female 43-year-old high school teacher in the southern city of Marbella who had no pre-existing health conditions and who died of a cerebral haemorrhage.
Neither health authorities nor the clinic where she was admitted would confirm details, citing data protection laws.
Monday's decision to suspend all AstraZeneca shots came just hours after Germany, France and Italy announced similar moves linked to fears the vaccine could generate serious side effects such as blood clots which can cause swellings, heart attacks and haemorrhages.
In a statement, the AEMPS said the three cases might be linked to the "formation of blood clots in areas of the body where they are less common" without drawing firm conclusions.
Its investigators were "gathering more information and carrying out an exhaustive investigation to find out whether there was not only a causal link to the administration of the vaccine, but also a possible temporal link".
READ ALSO:
- AstraZeneca benefits outweigh risks, EMA says as EU orders 10 million more Pfizer doses
- IN STATS: How the AstraZeneca suspension is affecting Spain’s Covid vaccine rollout
Until the use of the AstraZeneca vaccine was suspended, 975,661 people in Spain had received a shot, official figures show.
Spain, which is also administering the Pfizer/BioNtech and Moderna vaccines, has so far recorded more than 72,500 coronavirus deaths from more than 3.2 million cases.
For now, the European Medicines Agency (EMA) has said it is "firmly convinced" that benefits of AstraZeneca's vaccine outweigh potential risks, insisting there was no evidence linking it to blood clots.
Its experts are nonetheless looking into "adverse events" associated with all vaccines, and the regulator is due to publish its conclusions on Thursday.
The World Health Organization said Wednesday its experts were still reviewing safety data on the AstraZeneca Covid-19 vaccine following concerns around blood clots but recommended that injection programmes continue.
"The WHO Global Advisory Committee on Vaccine Safety is carefully assessing the latest available safety data," the UN health agency said in a statement.
"At this time, WHO considers that the benefits of the AstraZeneca vaccine outweigh its risks and recommends that vaccinations continue."
The global health body added that in mass vaccination campaigns, it was routine for countries to flag up potential adverse events after people have been immunised.
"This does not necessarily mean that the events are linked to vaccination itself, but it is good practice to investigate them. It also shows that the surveillance system works and that effective controls are in place," it said.
"Vaccination against Covid-19 will not reduce illness or deaths from other causes.
"Thromboembolic events are known to occur frequently. Venous thromboembolism is the third most common cardiovascular disease globally."
Comments
See Also
The announcement by Spain's AEMPS medicines agency came two days after the government suspended use of the vaccine for at least a fortnight as a precautionary measure.
The three cases occurred between late Monday and early Tuesday.
Local press reports said the person who died was a female 43-year-old high school teacher in the southern city of Marbella who had no pre-existing health conditions and who died of a cerebral haemorrhage.
Neither health authorities nor the clinic where she was admitted would confirm details, citing data protection laws.
Monday's decision to suspend all AstraZeneca shots came just hours after Germany, France and Italy announced similar moves linked to fears the vaccine could generate serious side effects such as blood clots which can cause swellings, heart attacks and haemorrhages.
In a statement, the AEMPS said the three cases might be linked to the "formation of blood clots in areas of the body where they are less common" without drawing firm conclusions.
Its investigators were "gathering more information and carrying out an exhaustive investigation to find out whether there was not only a causal link to the administration of the vaccine, but also a possible temporal link".
READ ALSO:
- AstraZeneca benefits outweigh risks, EMA says as EU orders 10 million more Pfizer doses
- IN STATS: How the AstraZeneca suspension is affecting Spain’s Covid vaccine rollout
Until the use of the AstraZeneca vaccine was suspended, 975,661 people in Spain had received a shot, official figures show.
Spain, which is also administering the Pfizer/BioNtech and Moderna vaccines, has so far recorded more than 72,500 coronavirus deaths from more than 3.2 million cases.
For now, the European Medicines Agency (EMA) has said it is "firmly convinced" that benefits of AstraZeneca's vaccine outweigh potential risks, insisting there was no evidence linking it to blood clots.
Its experts are nonetheless looking into "adverse events" associated with all vaccines, and the regulator is due to publish its conclusions on Thursday.
The World Health Organization said Wednesday its experts were still reviewing safety data on the AstraZeneca Covid-19 vaccine following concerns around blood clots but recommended that injection programmes continue.
"The WHO Global Advisory Committee on Vaccine Safety is carefully assessing the latest available safety data," the UN health agency said in a statement.
"At this time, WHO considers that the benefits of the AstraZeneca vaccine outweigh its risks and recommends that vaccinations continue."
The global health body added that in mass vaccination campaigns, it was routine for countries to flag up potential adverse events after people have been immunised.
"This does not necessarily mean that the events are linked to vaccination itself, but it is good practice to investigate them. It also shows that the surveillance system works and that effective controls are in place," it said.
"Vaccination against Covid-19 will not reduce illness or deaths from other causes.
"Thromboembolic events are known to occur frequently. Venous thromboembolism is the third most common cardiovascular disease globally."
Join the conversation in our comments section below. Share your own views and experience and if you have a question or suggestion for our journalists then email us at [email protected].
Please keep comments civil, constructive and on topic – and make sure to read our terms of use before getting involved.
Please log in here to leave a comment.